
The early cold cathode vacuum tubes, called Crookes tubes, used a high electrical potential between the anode and the cathode to ionize the residual gas in the tube. To release electrons into the tube, they must first be detached from the atoms of the cathode. The image in a classic television set is created by focused beam of electrons deflected by electric or magnetic fields in cathode ray tubes (CRTs).Ĭathode rays are so named because they are emitted by the negative electrode, or cathode, in a vacuum tube. Electrons were first discovered as the constituents of cathode rays. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, the glass opposite the negative electrode is observed to glow from electrons emitted from the cathode. crookes tube: An early experimental electrical discharge tube, invented by English physicist William Crookes and others around 1869-1875, in which cathode rays, streams of electrons, were discoveredĬathode rays (also called electron beams or an e-beams) are streams of electrons observed in vacuum tubes.cathode rays: Streams of electrons observed in vacuum tubes.Thomson used the cathode ray tube to determine that atoms had small negatively charged particles inside of them, which he called “electrons.” Cathode rays carry electronic currents through the tube.
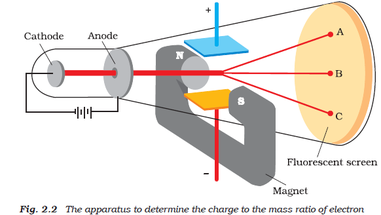
Researchers realized that something was traveling from the anode when objects placed in the tube in front of it could cast a shadow on the glowing wall.Electrons accelerated to high velocities travel in straight lines through an empty cathode ray tube and strike the glass wall of the tube, causing excited atoms to fluoresce or glow.Everett who helped to greatly increase Thomson's experimental range. About 1894 he acquired an excellent glassblower named E. He was very fumble fingered and had a tendancy to break things. Incidently, Thomson was a very unhandy person. The amount the cathode ray bent from the straight line using either the electric field or the magnetic field allowed Thomson to calculate the e/m ratio. This allowed him to use either electrical or magnetic or a combination of both to cause the cathode ray to bend. Thomson also could use magnets, which were placed on either side of the straight portion of the tube just to the right of the electrical plates. The two plates about midway in the CRT were connected to a powerful electric battery thereby creating a strong electrical field through which the cathode rays passed. The long glass finger (in the photo) projecting downward from the right-hand globe is where the entire tube was evacuated down to as good as a vacuum as could be produced, then sealed. Thomson in 1897 announcing the discovery of the electron. Th diagram below appeared in an article by J.J. It is about one meter in length and was made entirely by hand. The image below of a CRT used by Thomson in his experiments. Only the end of the CRT can be seen to the right-hand side of the picture. Thomson and a cathode ray tube from around 1897, the year he announced the discovery of the electron. Thomson used results from cathode ray tube (commonly abbreviated CRT) experiments to discover the electron.
